70以上 s p d f orbitals periodic table 465434-S p d f orbital blocks periodic table
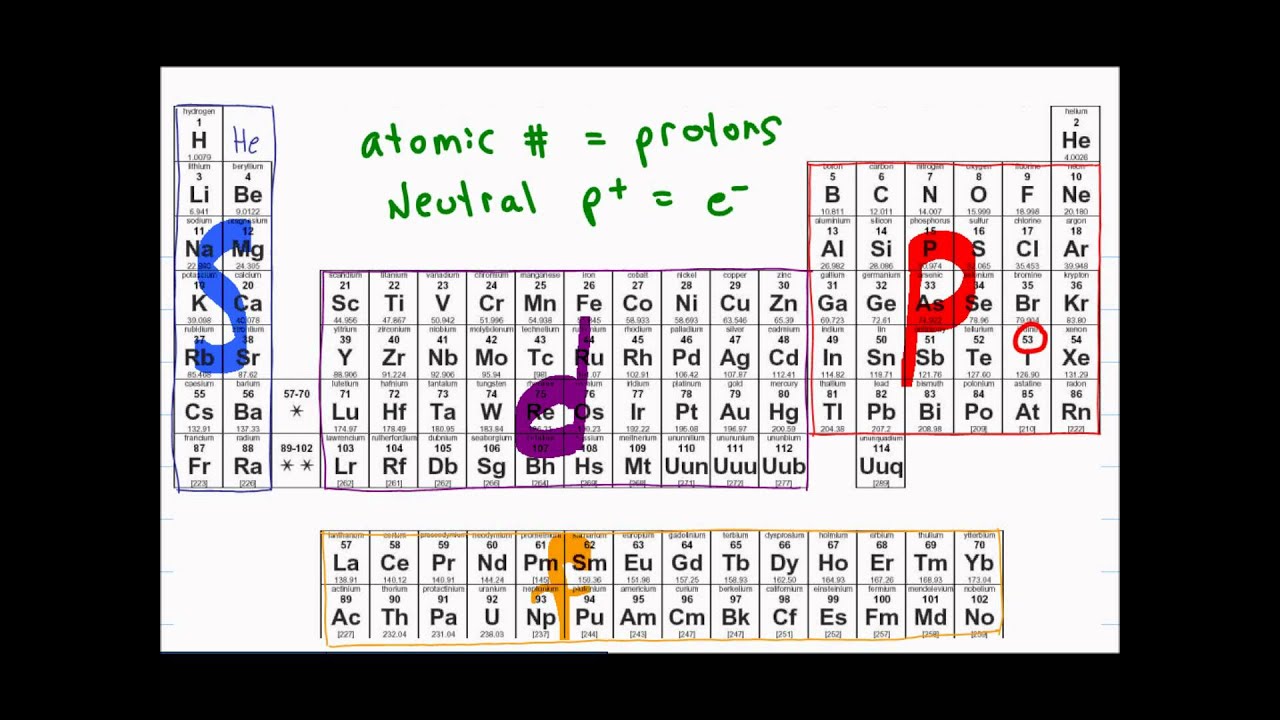
The periodic table, electron shells, and orbitals Shells, subshells, and orbitals This is the currently selected item Introduction to electron configurations subshell, or sometimes people will say sublevels and that's where they're talking about s or p or d and eventually f so if I circle this, I'm talking about that first shell Now2s is lower energy than 2p)(image source)So for example,The seven f orbitals in the 4n electron shell, together with the single s orbital, three p orbitals and four d orbitals, allow it to contain a total of thirtytwo (32) electrons The geometry of the f orbitals is more complex than any of the previous orbitals we have looked at

7 3 Electron Configurations Of Atoms Chemistry Libretexts
S p d f orbital blocks periodic table
S p d f orbital blocks periodic table-Explore the Bohr model and atomic orbitals Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity If you're seeing this message, it means we're having trouble loading external resources on our websiteExplore the Bohr model and atomic orbitals Learn how to use an element's position on the periodic table to predict its properties, electron configuration, and reactivity If you're seeing this message, it means we're having trouble loading external resources on our website


Ib Chemistry Topic 12 1 Electronic Configuration
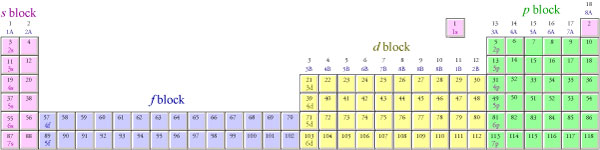
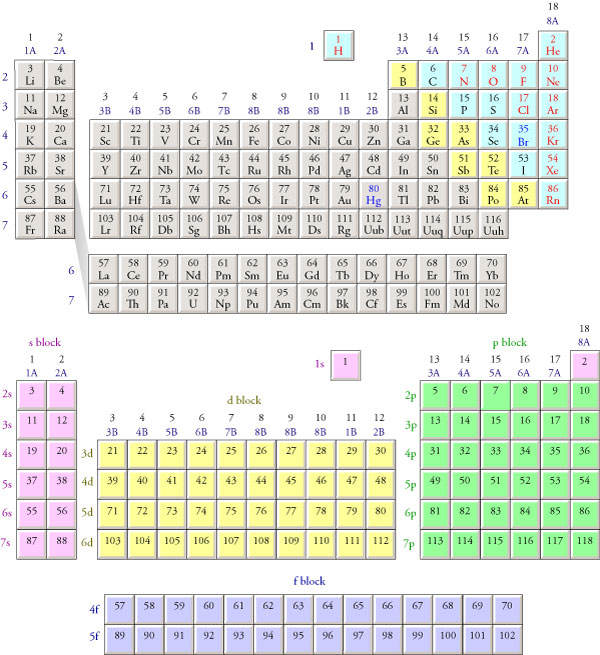
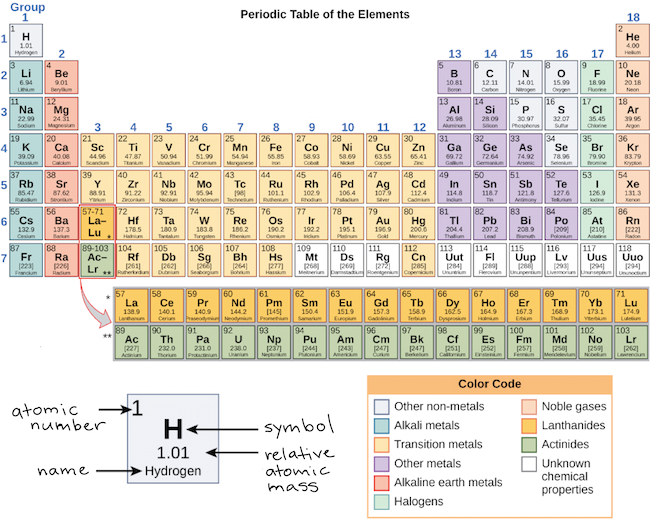
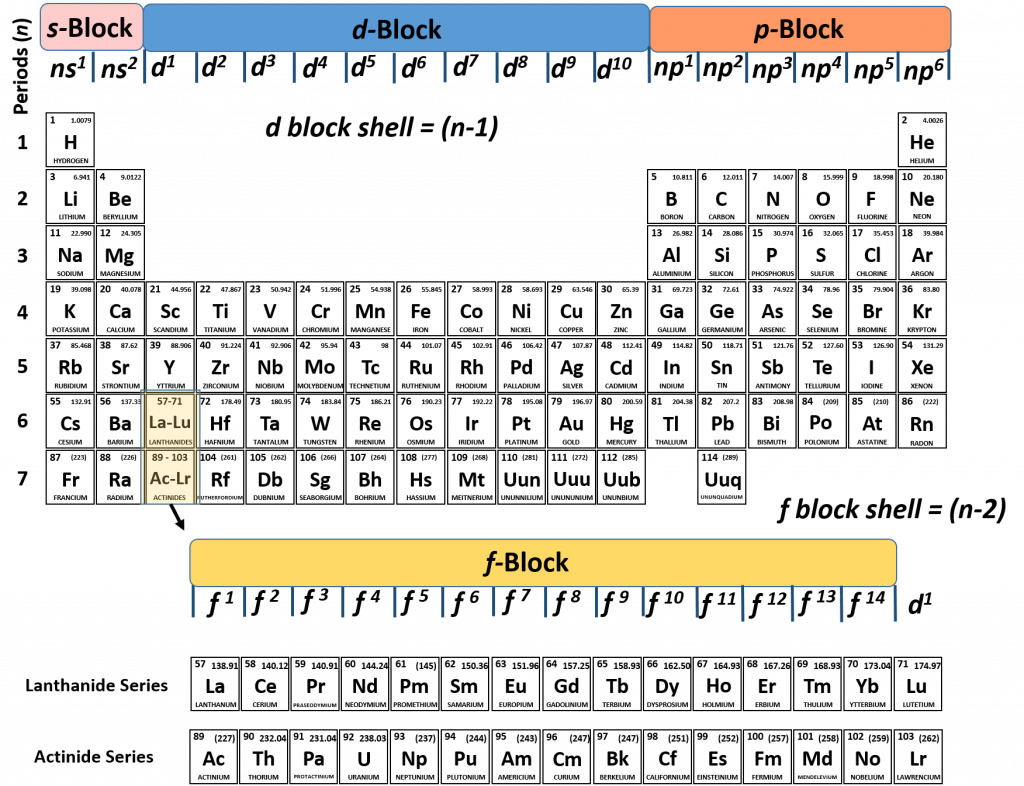
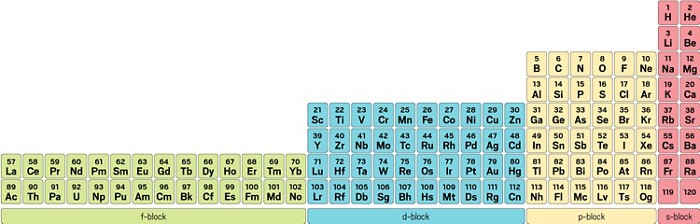
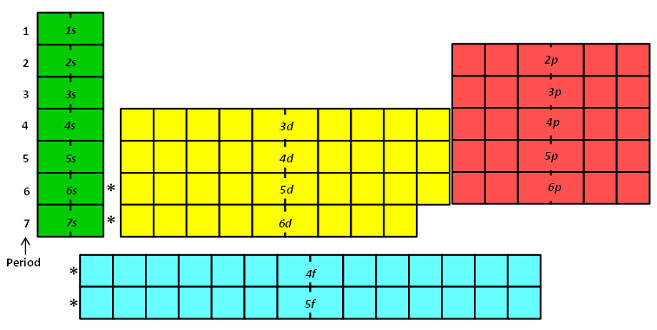
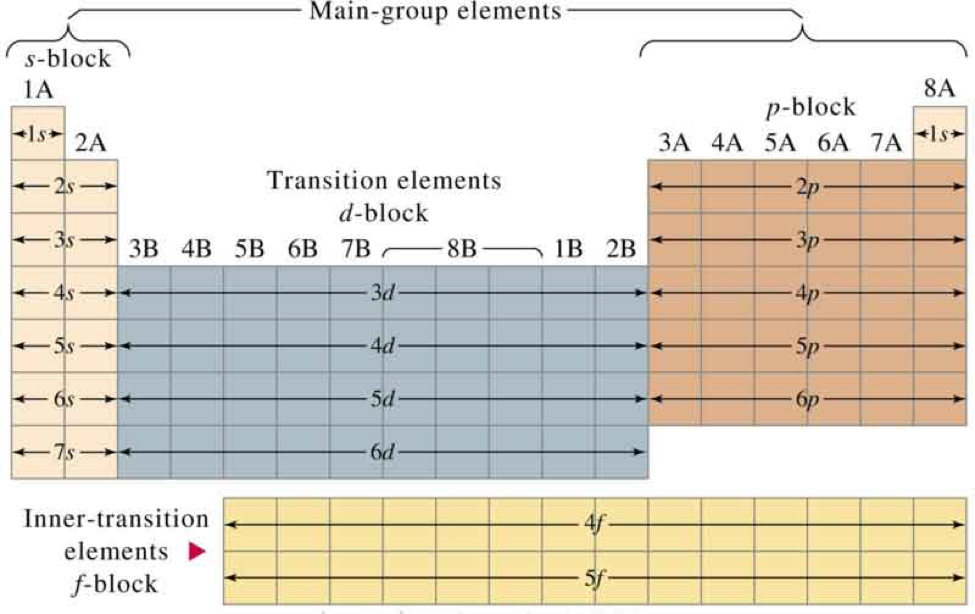
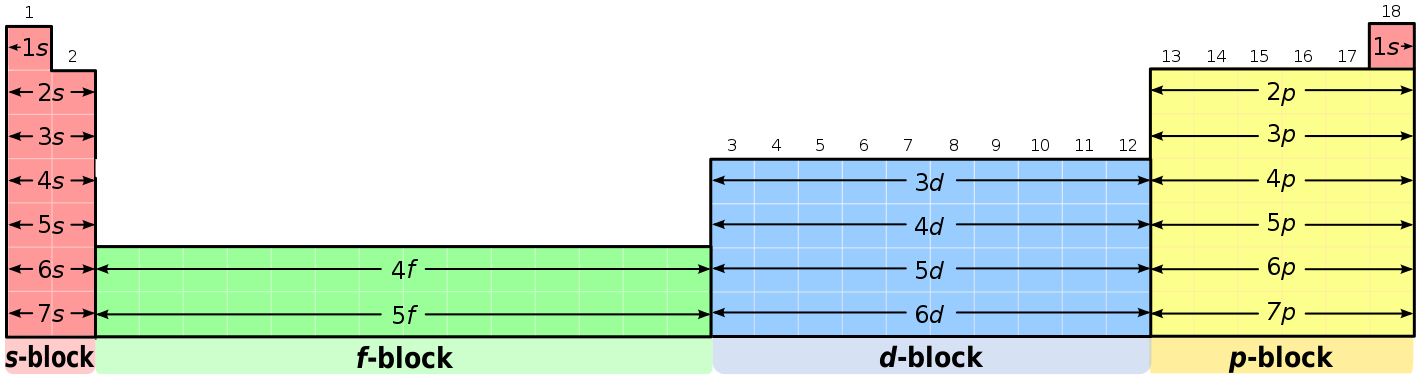
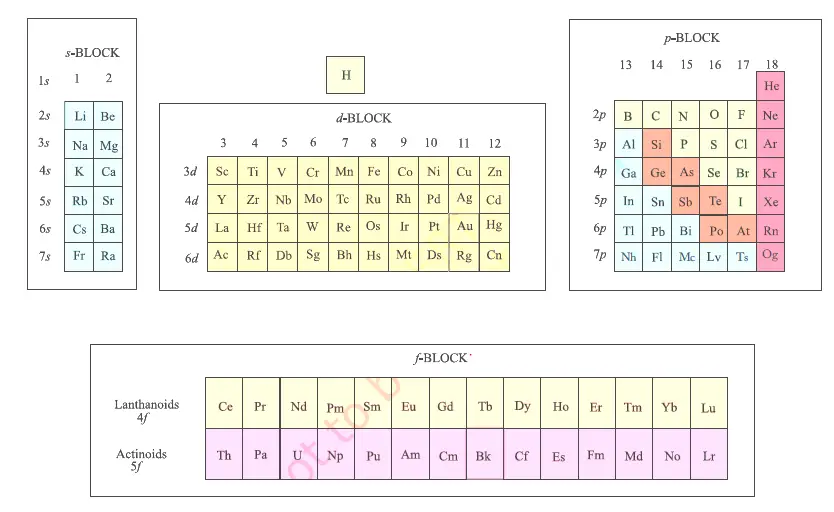
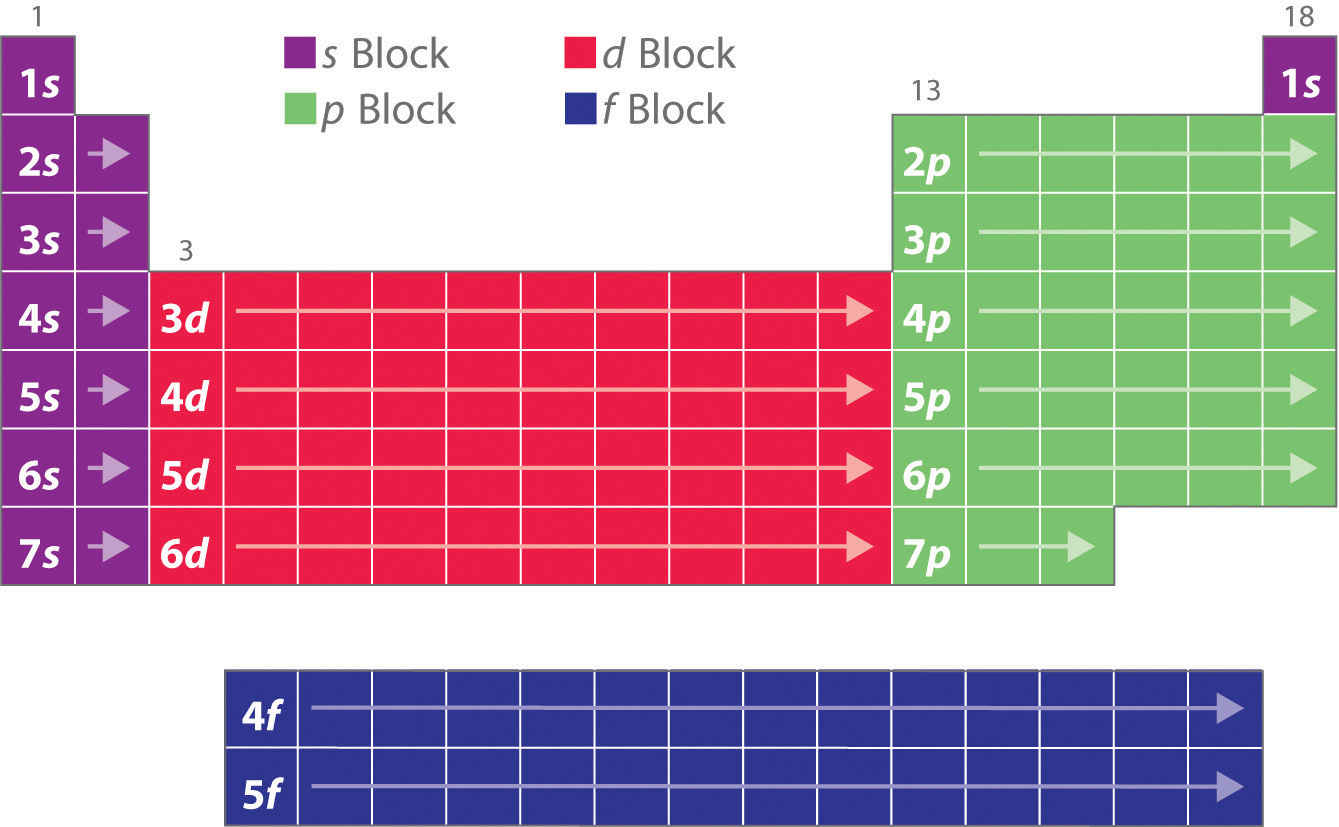
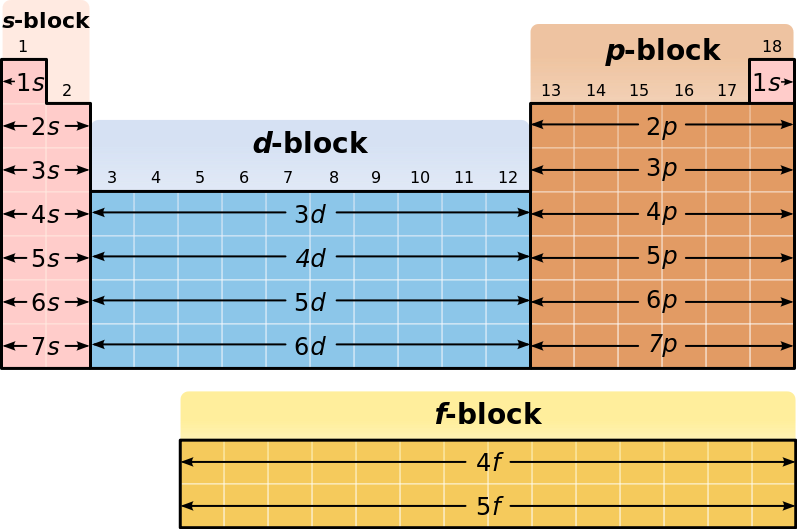
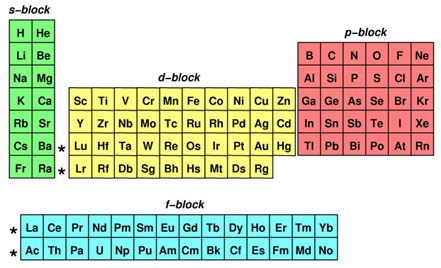
This cloud takes different shapes depending on the energy level of the electron, with a sequence described in the Periodic Table of Elements These are the s, p, d and f blocks of atomic elements and their shapes Orbital Shapes (s, p, d and f)The periodic table shows us the sequential filling of the electrons The energy of the orbitals determines the sequence of filling Lower energy orbitals are always preferred over high energy onesThe table is thus divided into 4 blocks namely – s,p,d, f blocks, depending on the occupation of the respective orbitals by the valence electrons of an elementThe term appears to have been first used by Charles Janet Each block is named after its characteristic orbital sblock, pblock, dblock, and fblock A long periodic table showing, from left to right the s, d, f, and pblocks The fblock, normally shown as a footnote, here is placed between the s and dblocks
The actinides and lanthanides have been placed at the bottom of the periodic table to avoid the undue expansion of the periodic table 1 I st inner transition or 4 fseries, contains 14 elements 58 Ce to 70 Lu Filling of electrons takes place in 4f subshell 2 IInd inner transition or 5 fseries, contains 14 elements 90 Th to 103 Lr FillingThis video explains s, p, d, and f orbitals, sublevels, and their shapes It discusses the 4 quantum numbers n, l, ml, and ms n represents the energy leve18 Ar Argon
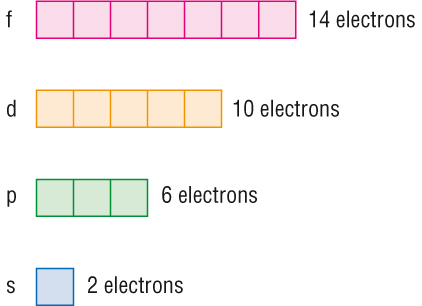
These orbitals are designated as s, p, d, f among othersThe energy incre Snehapareek277 Snehapareek277 Chemistry Secondary School Variations of s p d and f orbital energies of atoms in the periodic table 1 See answer Snehapareek277 is waiting for your help Add your answer and earn points17 Cl Chlorine 3545;Maximum 6 electrons in 3 orbitals Maximum 2 electrons in 1 orbital Maximum 10 electrons in 5 orbitals Maximum 14 electrons in 7 orbitals



Oneclass Write The Electron Configurations For P And Cl Using Both Spdf Notation And Orbital Box Dia



7 3 Electron Configurations Of Atoms Chemistry Libretexts
Classification of Elements and Periodicity in Properties homework assignment help is most useful online help portal for the students that providing all Online spdf Block Elements assignment help Services In the long form of the periodic table, elements are grouped into four main blocks, purely on the basis of electronic configurations Elements are grouped in blocks 's', 'p', 'd' and 'f' depending on the nature of orbital (s) into which the last electron of the atom entersIn the above diagram, the boxes refer to elements and not to orbitals directly For example, the oxygen atom, which contains four electrons in 2p orbitals, is placed in the fourth box in the 2p group If you think about it, this table provides a beautiful view of how the arrangement of the periodic table is a direct consequence of the number of orbitals of each type and their relative energiesS p d f g The s, p, d, f and g are called atomic orbitals Filling up these orbitals with electrons builds atoms, and the way in which atoms are build up gives rise to the periodic table There is only one s orbital (m l = 0), but there are three p orbitals (m l = −1,0,1), five d orbitals (m l = −2,−1,0,1,2), and seven f orbitals (m
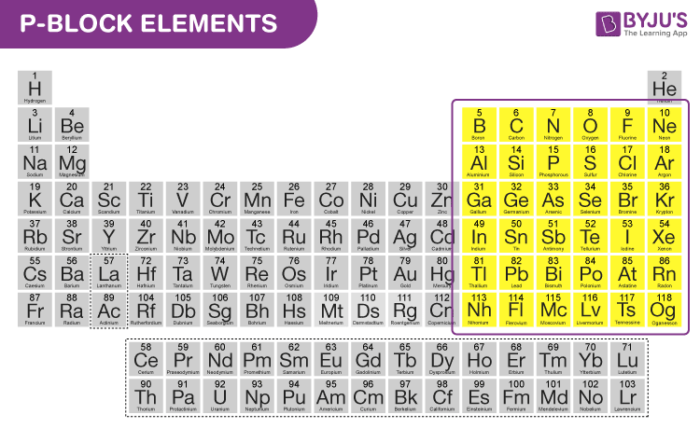


P Block Elements On Periodic Table Introduction Properties Trends


If Each Orbital Can Hold A Maximum Of 3 Electrons The Number Of Elements In The 4th Period Of The Periodic Table Long Form Is Socratic
The p subshell (dumbbell orbital) has 3 porbitals, so it can contain 6 electrons max The d subshell has 5 dorbitals, so it can contain 10 electrons max And the f subshell has 7 forbitals, so it can contain 14 electrons max The first shell has one subshell an s subshellMaximum 6 electrons in 3 orbitals Maximum 2 electrons in 1 orbital Maximum 10 electrons in 5 orbitals Maximum 14 electrons in 7 orbitalsS, p, d, f and so on are the names given to the orbitals that hold the electrons in atoms These orbitals have different shapes (eg electron density distributions in space) and energies (eg 1s is lower energy than 2s which is lower energy than 3s;


Electron Configuration Of Atoms Shortcut Tutorial Video Youtube


Electron Configurations
S, p, d, and f The different sections of the Periodic Table are very important in understanding Electron Configuration There are 4 "Blocks" in the Periodic Table the sblock, pblock, dblock, & fblock Remember the special rules for the d and f blocks d – n1 f – n 2This video explains s, p, d, and f orbitals, sublevels, and their shapes It discusses the 4 quantum numbers n, l, ml, and ms n represents the energy leveMagnetism and Electronic Configurations The distribution of electrons in an atom, molecule, or ion in atomic or molecular orbitals Electrons can be in s,p,d, or f orbital Shells


Electron Configurations


The Periodic Table Electron Shells And Orbitals Article Khan Academy
Electron Configuration s, p, d, and f The different sections of the Periodic Table are very important in understanding Electron Configuration There are 4 "Blocks" in the Periodic Table the sblock, pblock, dblock, & fblockThe periodic table consists of four blocks of elements that correspond to s, p, d, and f orbitals being filled After f orbitals come g and h orbitals In theory, if a g block and an h block of elements existed, bow long would the rows of g and h elements be in this theoretical periodic table?16 S Sulfur 36;



Modern Periodic Table S P D F Blocks Elements
/800px-Orbital_representation_diagram.svg-589bd6285f9b58819cfd8460.png)


Electron Configuration Chart
15 P Phosphorus ;Corresponds to the block grouping s,p,d,f on the Periodic Table orbitals regions within electron cloud where electrons orbit the nucleus;Periodic table 8b why 3 columns Tutorial on s,p,d,f orbitals picture of periodic table with d block what is the 2p block periodic table why there are three 8b elements in periodic table show images of periodic table showing mass and atomic numbers periodic table visual basic pblock elementstutorial "pblock" chemistrytutorial The four main



Modern Periodic Table Of Elements Periodicity Trends Videos Examples



3 1 S P D F Periodic Table Kerem S Chemistry Notes Ib
Can hold a maximum of 2 electrons;"s" subshell One possible orientation "p" subshell Three possible orientations There are five possible orbitals in a "d" subshell, and 7 possible orbitals in an "f" subshell!332 Identify the organization of the periodic table into energy levels, sublevels (s, p, d, and f), and orbitals Using the periodic table, write electron configurations for atoms up to Z=18 Identify the repeating pattern of electron configurations for elements in a group



Periodic Table With Spdf Configuration Of All Elements Periodic Table Electron Configuration Configuration


Www Gusd Net Cms Lib Ca Centricity Domain 1907 6the periodic table 3 5web Pdf
The s, p, and dblock elements of the periodic table are arranged into 18 numbered columns, or groups The elements in each group have the same number of valence electrons As a result, elements in the same group often display similar properties and reactivityThe labels s, p, d and f blocks of the Periodic Table refer to the subshell that is being filled with electrons Group 1 elements occur at the beginning of a new row (Period) of the Periodic Table The highest energy level (valence shell) contains only 1 electron in an s subshell Group 2 elements occur directly to the right of Group 1 elementsIn the above diagram, the boxes refer to elements and not to orbitals directly For example, the oxygen atom, which contains four electrons in 2p orbitals, is placed in the fourth box in the 2p group If you think about it, this table provides a beautiful view of how the arrangement of the periodic table is a direct consequence of the number of orbitals of each type and their relative energies



Electron Configurations Using Periodic Table Explained In Easy Tutorial Crash Chemistry Academy Youtube


Electronic Configuration And The Periodic Table Table Chemistry For Class 11 In Hindi Youtube
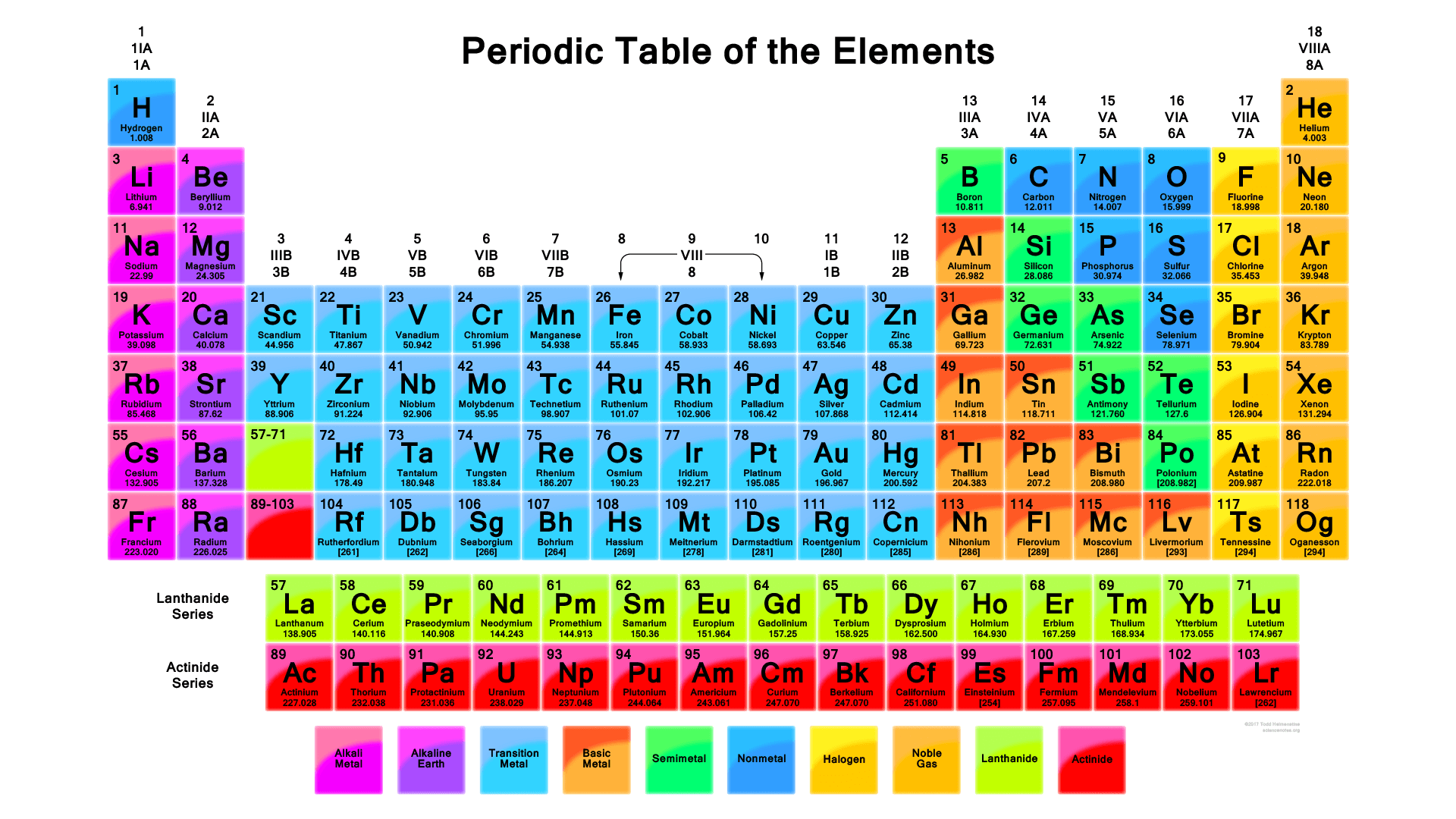
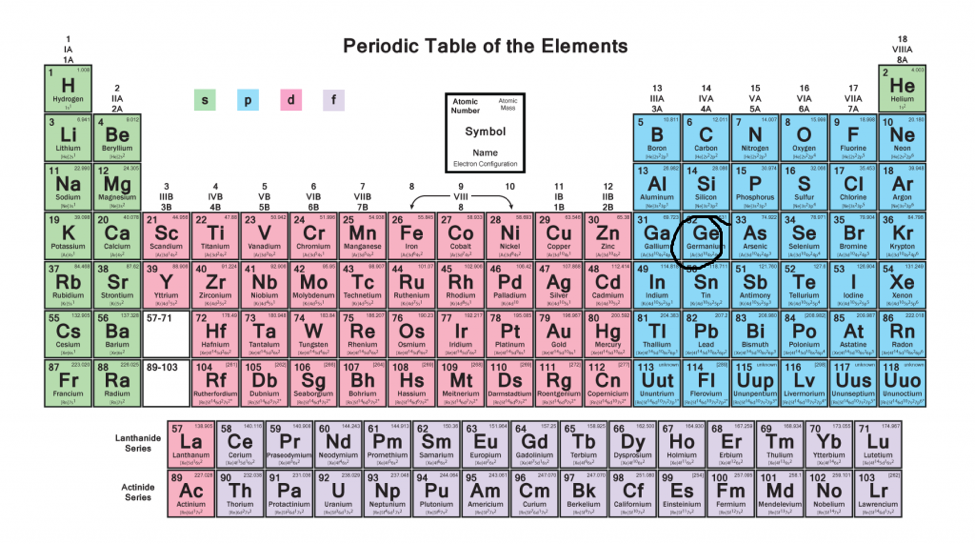
Fred Senese of Antoine Frostburg explains "You might expect that the 's' stands for 'spherical' and 'p' stands for 'polar' because these imply the shapes of the s and p orbitals, but unfortunately, the letter designations have nothing to do with1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f s can hold 2 electrons p can hold 6 electrons d can hold 10 electrons f can hold 14 electrons Note that individual orbitals hold a maximum of two electrons There can be two electrons within an s orbital, p orbital, or d orbitalThe elements of the Periodic table are also classified block wise There are 4 blocks in the Periodic table s block, p block, d block and f block The elements which are in the s block have the valence electrons in sorbitals Similarly, elements which are in the p block have the valence electrons in porbitals


The Periodic Table Is An Icon But Chemists Still Can T Agree On How To Arrange It


Electron Configurations
4 blocks of Periodic table The elements of the Periodic table are also classified block wise There are 4 blocks in the Periodic table s block, p block, d block and f block The elements which are in the s block have the valence electrons in sorbitalsThis is a video tutorial on s, p, d, f orbitals and their location in the periodic table that are called blocks This will also help in electron configurationThe block names (s, p, d, f) originated from descriptions of spectroscopic lines of atomic orbitals sharp, principal, diffuse, and fundamental No gblock elements have been observed to date, but the letter was chosen because it is next in alphabetical order after f



Electron Configurations Atomic Properties And The Periodic Table



Periodic Table Spdf Blocks Page 1 Line 17qq Com
The orbitals are filled in the order 1 s, 2 s, 2 p, 3 s, 3 p, 4 s, 3 d, 4 p, 5 s, 4 d, 5 p, 6 s, 4 f, 5 d, 6 p, 7 s, 5 f Each orbital can hold no more than two electrons Two electrons in the same orbital must have opposite spins (the Pauli exclusion principle)Periodic table 8b why 3 columns Tutorial on s,p,d,f orbitals picture of periodic table with d block what is the 2p block periodic table why there are three 8b elements in periodic table show images of periodic table showing mass and atomic numbers periodic table visual basic pblock elementstutorial "pblock" chemistrytutorial The four mainThe position of an element in the periodic table is based on its valence shell electronic configuration Based on the type of atomic orbitals which receives the last electron or differentiating electron the elements are categorized as s, p, d and fblock elements There are two exceptions to this classification The first exception is Helium


Q Tbn And9gctvfpxbcvo7zlvnrm1sxwkevdw4glufh 1wmgdzr91s1kn63ryc Usqp Cau



Free Printable Periodic Tables Pdf And Png Science Notes And Projects
You will see the lowercase letters s, p, d, f, g, and h for the suborbitals For example, the electron in a hydrogen (H) atom would have the values n=1 and l=0 The single electron would be found in the "K" shell and the "s" suborbital If you go on to learn more about chemistry, you may see its description written as 1s1Shapes of Orbitals and Electron Density Patterns The s orbitals are spherical, while p orbitals are polar and oriented in particular directions (x, y, and z) It may be simpler to think of these two letters in terms of orbital shapes (d and f aren't described as readily)However, if you look at a crosssection of an orbital, it isn't uniformExplore each Elements orbitals and electron configuration View all 118 elements in one interactive Periodic Table Breaking Atom About us Periodic Table States Orbitals Electronegativity Evolution Games Learn Calculators Get Help Elements Glossary Contact Boiling Point Melting Point Elements


Electron Configurations How To Write Out The S P D F Electronic Arrangements Of Atoms Ions Periodic Table Oxidation States Using Orbital Notation Gce A Level Revision Notes



Periodic Table Blocks Of Elements
The orbitals are s, p, d, and f How many electrons can each orbital hold?For the first eighteen elements in the periodic table up to and including argon (ie the first three periods of the table), all of the electrons will be found in either s orbitals or p orbitals In fact, that continues to be the case for the first two elements of period four potassium and calciumA block of the periodic table is a set of elements unified by the orbitals their valence electrons or vacancies lie in The term appears to have been first used by Charles Janet Each block is named after its characteristic orbital sblock, pblock, dblock, and fblock


Ch150 Chapter 2 Atoms And Periodic Table Chemistry



Powerpoint Orbital Shape Orientation Spdf Periodic Table Powerpoint Presentation Free Online Download Ppt 6tz333
The maximum electrons that can be carried by the subshell S is 2, by P is 6, by D is 10, and the F subshell can carry 14 This decides the electron capacity of the shells The K shell contains a 1s subshell hence it can carry 2 electrons, the L shell has 2s and 2p, and can carry 8 electronsPeriodic table 8b why 3 columns Tutorial on s,p,d,f orbitals picture of periodic table with d block what is the 2p block periodic table why there are three 8b elements in periodic table show images of periodic table showing mass and atomic numbers periodic table visual basic pblock elementstutorial "pblock" chemistrytutorial The four mainNumber of orbitals differs with sublevel type (s,p,d,f)


The Periodic Table Is An Icon But Chemists Still Can T Agree On How To Arrange It


Electron Configuration Texas Gateway
The s sublevel has one orbital, the p sublevel has three orbitals, the d sublevel has five orbitals, and the f sublevel has seven orbitals In the first period, only the 1 s sublevel is being filled Since all orbitals can hold two electrons, the entire first period consists of just two elementsRegions within the energy levels;"s" subshell One possible orientation "p" subshell Three possible orientations There are five possible orbitals in a "d" subshell, and 7 possible orbitals in an "f" subshell!


Electron Configurations A Must Know Hack


Q Tbn And9gcrzchn Z2jxrzjr5karsdy96xglwxheyj J2iq56dlkjh9mytuq Usqp Cau
2 electrons What does "l" mean orbital type Describes the shape of the orbital (s, p, d, f) What does mL mean?(quantum number) specific orbital Meaning, it determines how many orbitals there are of a type per energy level (middle section of the periodic tableS has one orbital and can hold 2 electrons P has three orbitals and holds 6 electrons D 5 orbitals, 10 electrons F 7 orbitals and 14 electronsThe four different orbital forms (s, p, d, and f) have different sizes and one orbital will accommodate up to two electrons at most The orbitals p, d, and f have separate sublevels and will thus accommodate more electrons As shown, each element's electron configuration is unique to its position on the periodic table How do orbitals work?



Electron Configurations


File Periodic Table Blocks Spdf 32 Column Svg Wikimedia Commons



Periodic Table Spdf Part 2 Ground State Configuration Youtube


Q Tbn And9gct1uojy0cb9rtjch1agatf7uxadqzufdnxofdrpgkc 5nahzprw Usqp Cau



Ground State Electron Configuration Definition Example Video Lesson Transcript Study Com


Modern Periodic Table S P D F Blocks Elements


How Do You Determine The Electron Configuration Of W Socratic



Electron Configuration Basics Chemistrybytes Com


Parsing The Spdf Electron Orbital Model



Electron Configuration Anomalies Villanova College Chemistry Blog


Chapter 6 The Periodic Table And Periodic Law Mr Caldwell S Classroom



Periodic Table



Dublin Schools Lesson Electron Configurations Using The Periodic Table


Module B Lesson 4 1 T Gem And Chemland Mel Burgess And Met



File Subshells Of Orbitals Jpg Wikibooks Open Books For An Open World


6 9 Electron Configurations And The Periodic Table Chemistry Libretexts


5 6 Atomic Electron Configurations Chemistry Libretexts


Electron Configurations A Must Know Hack



Electronic Configuration Of Elements Trick S P D F Pattern Class 11 Iit Jee Neet Aiims Youtube


What Is The Electron Configuration For Francium Socratic



How To Find Elements Electron Configuration For Gold Au


Parsing The Spdf Electron Orbital Model


Parsing Spdf Orbital Hybridization And Simple Bonding


Ib Chemistry Topic 12 1 Electronic Configuration


Electron Configuration Texas Gateway



S Block Elements On The Periodic Table Properties Overview Video Lesson Transcript Study Com


Solved Select An Element On The Periodic Table To See Its Chegg Com


Electron Configurations
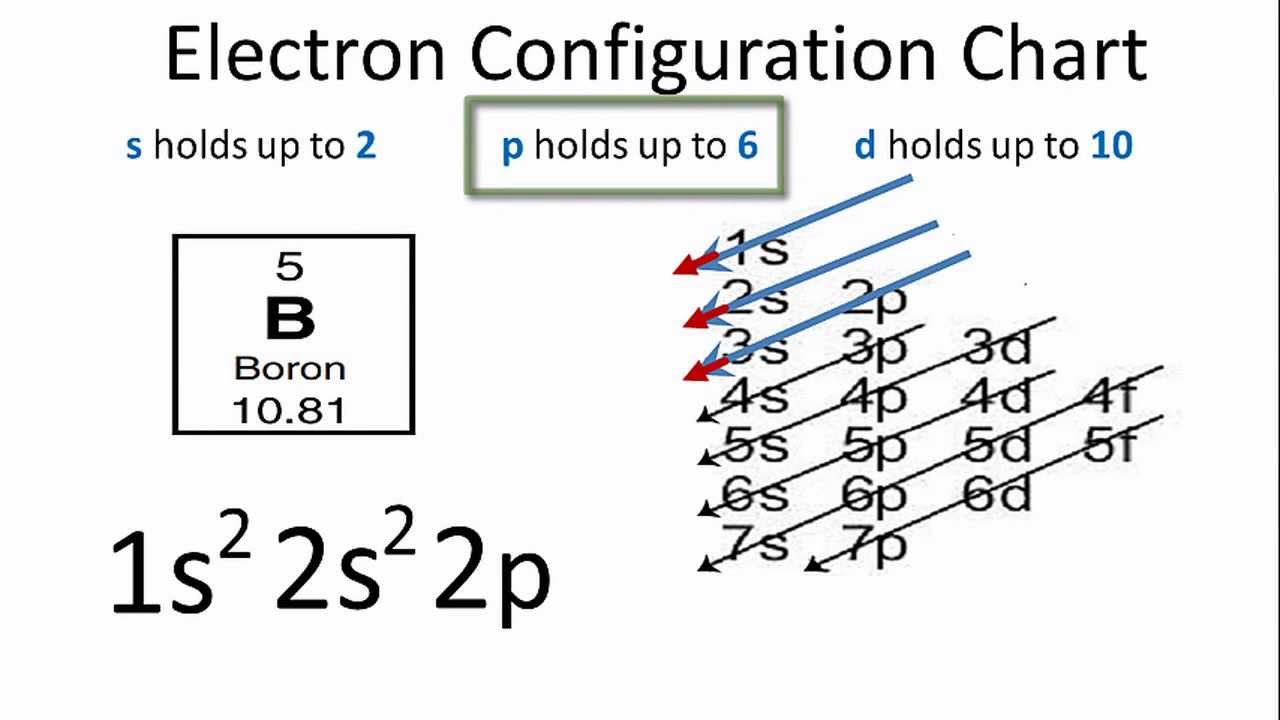


Electron Configuration For Carbon C
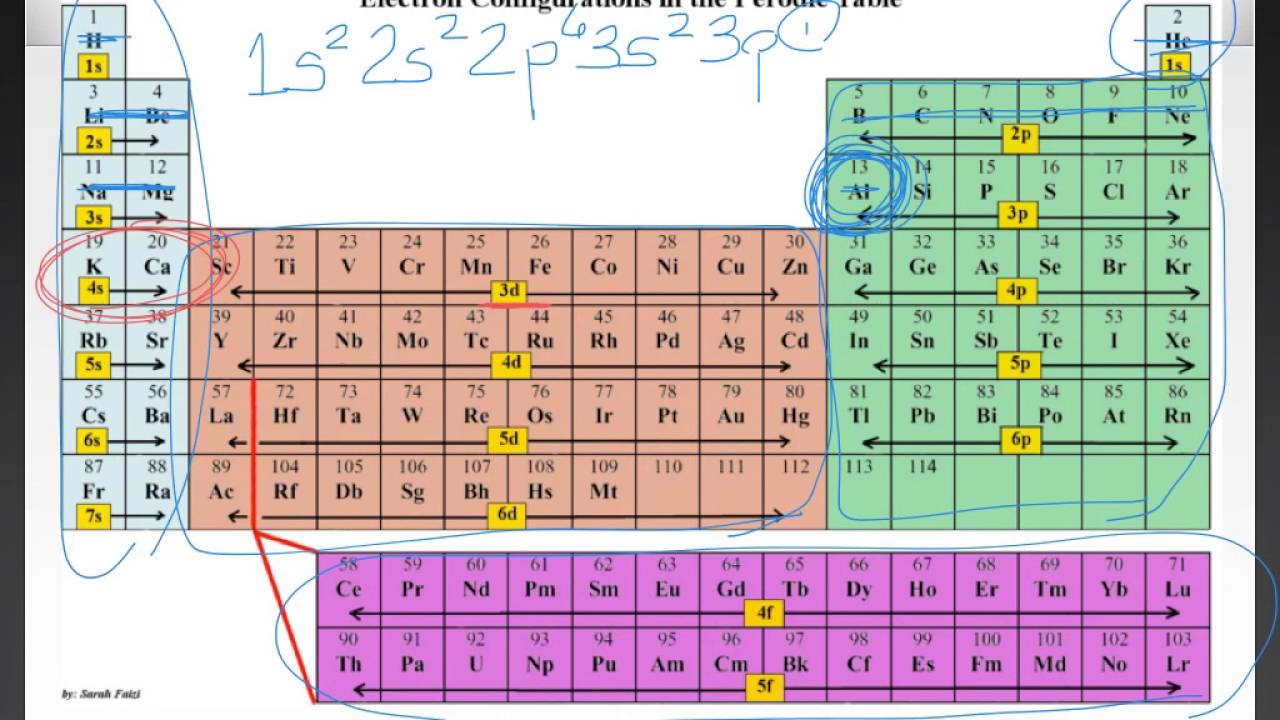


Electron Arrangement In Atoms Youtube



Valence Electron Definition Configuration Example Study Com Chemistry Lessons Chemistry Classroom Electron Configuration



The Actinide Research Quarterly 1st Quarter 04



Ib Chemistry On Quantum Numbers And Electronic Configuration
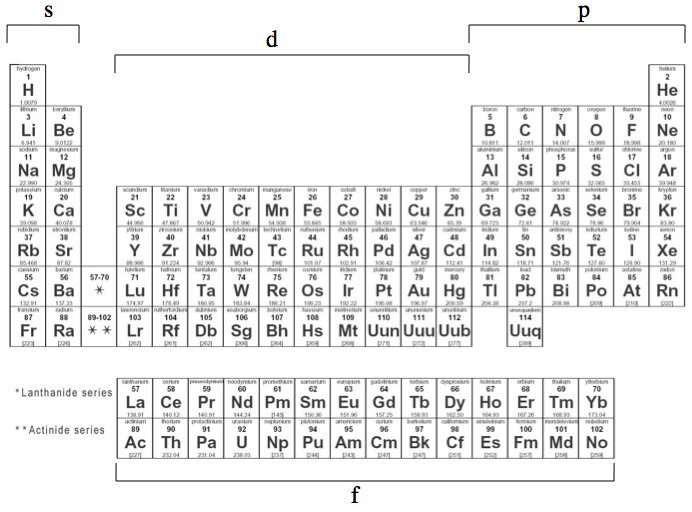


Q Is It Possible For An Atomic Orbital To Exist Beyond The S P F And D Orbitals They Taught About In School Like Could There Be A Other Letter Orbital Beyond


Electron Configurations And The Periodic Table
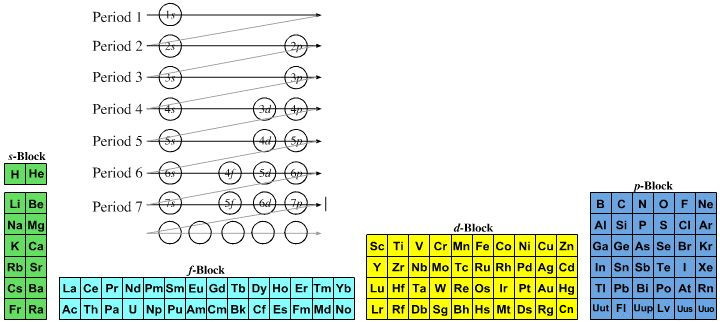


Quantum Numbers To Periodic Tables Chemogenesis
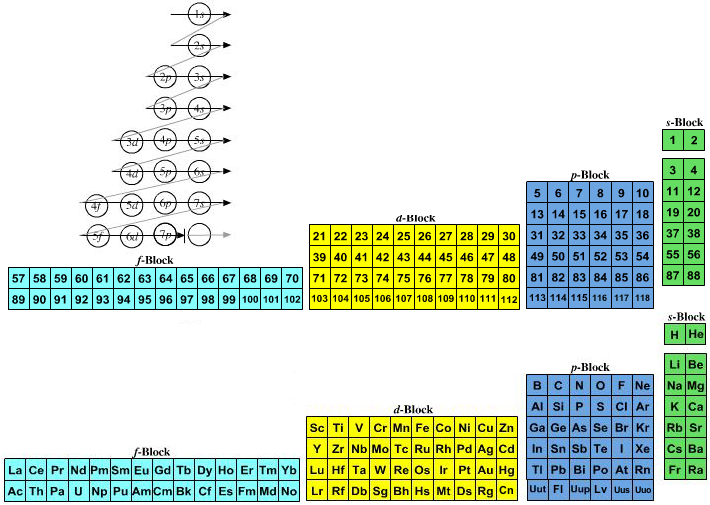


Quantum Numbers To Periodic Tables Chemogenesis


Www Ch Ntu Edu Tw Sfcheng Html Genchem05 Chapt6 Brown holme 2 Pdf
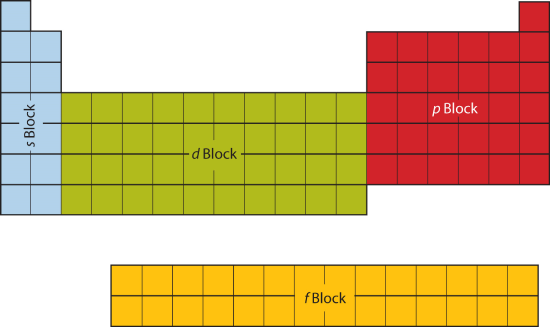


9 7 Electron Configurations And The Periodic Table Chemistry Libretexts
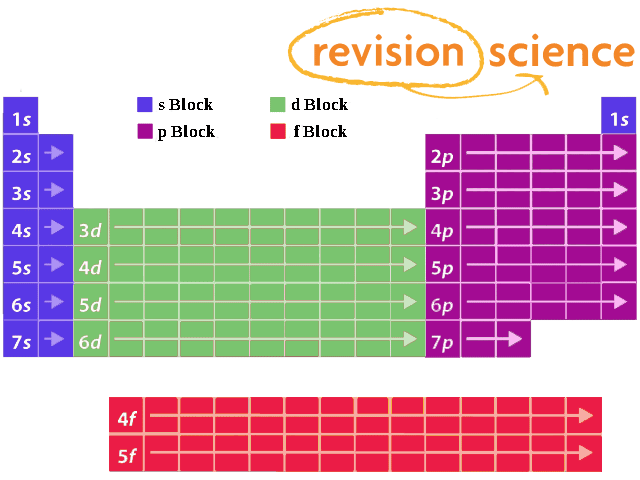


Shells And Subshells A Level Chemistry



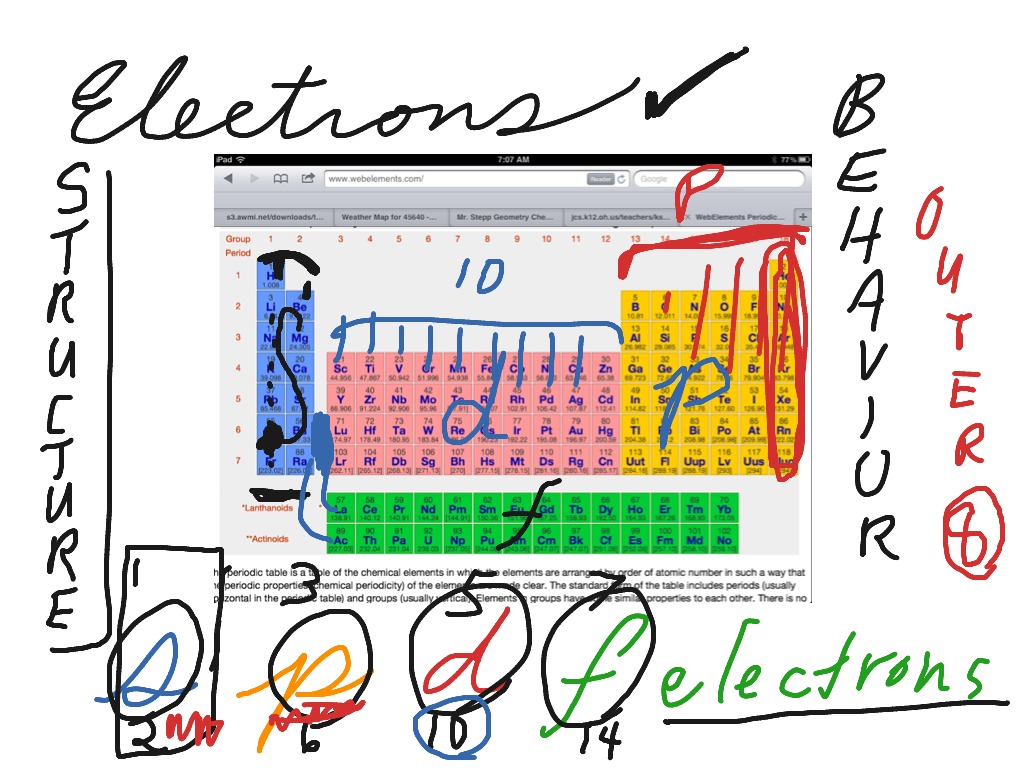
S P D F Electron Blocks On The Periodic Table Chemistry Periodic Table Electron Configuration Showme
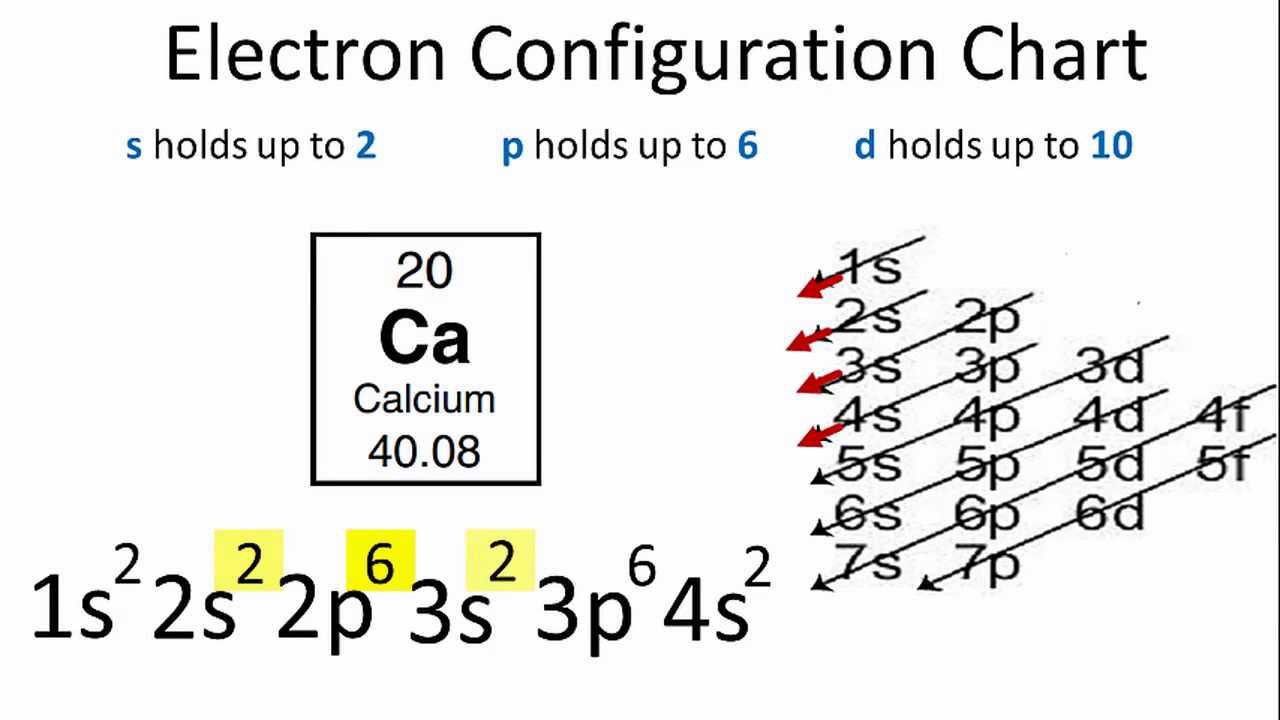


Electron Configuration For Calcium Ca



Blocks Of The Periodic Table Chemistry For Non Majors


Valence Electron Assignment Point


Q Tbn And9gcqgp28lgnr6xcufpgiroedaenthgrys Oedjaqmrwowa3gfhdra Usqp Cau


Parsing The Spdf Electron Orbital Model



Modern Periodic Table S P D F Blocks Periodic Table Timeline
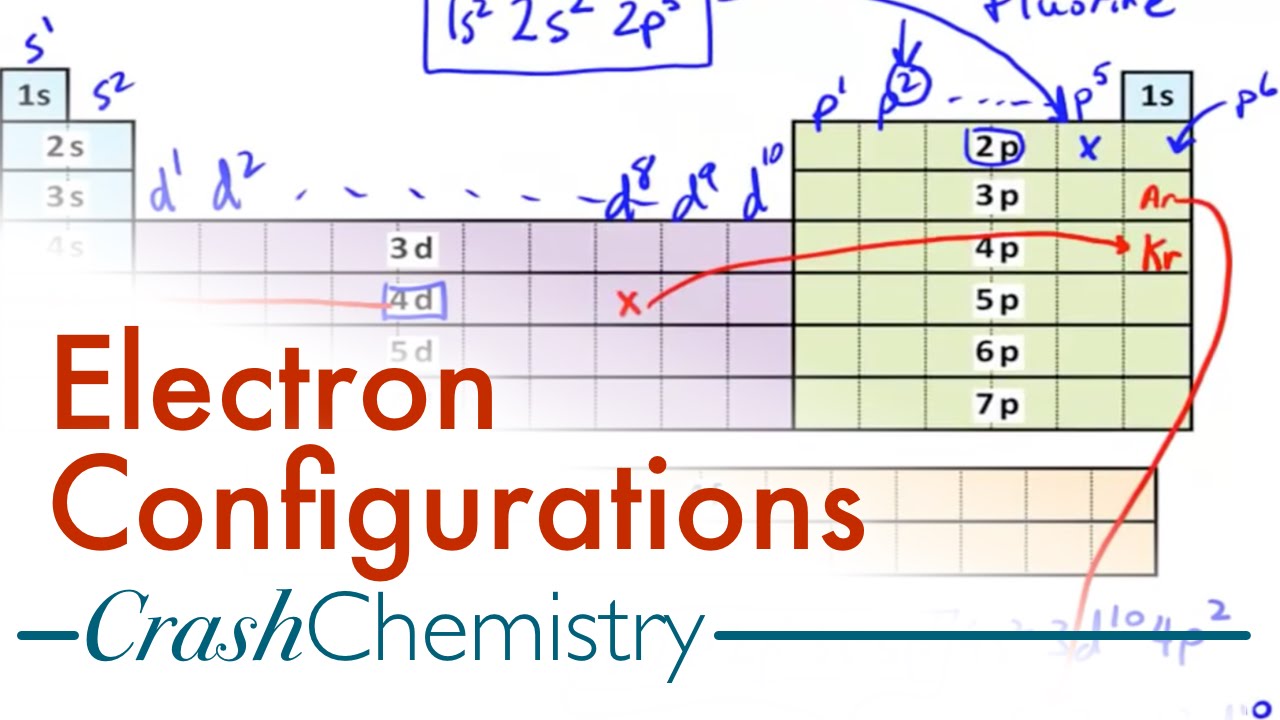


Electron Configuration Tutorial How To Derive Configurations From Periodic Table Crash Chemistry Youtube



Spdf Orbitals Location Diagram Quizlet



The Compound Interest Periodic Table Of Data Compound Interest



Electronic Configuration Of The D Block Elements Concepts Videos Q As


Blocks Of The Periodic Table
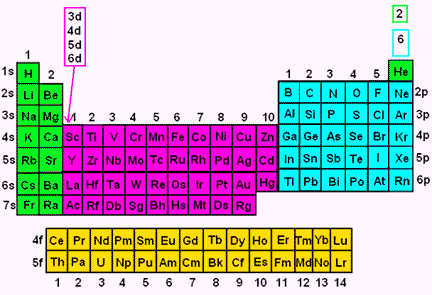


Electron Configuration Wyzant Resources



Which Orbital Indicated On The Periodic Table S P D F Help Asap I Ll Mark As Brainlister Brainly Com
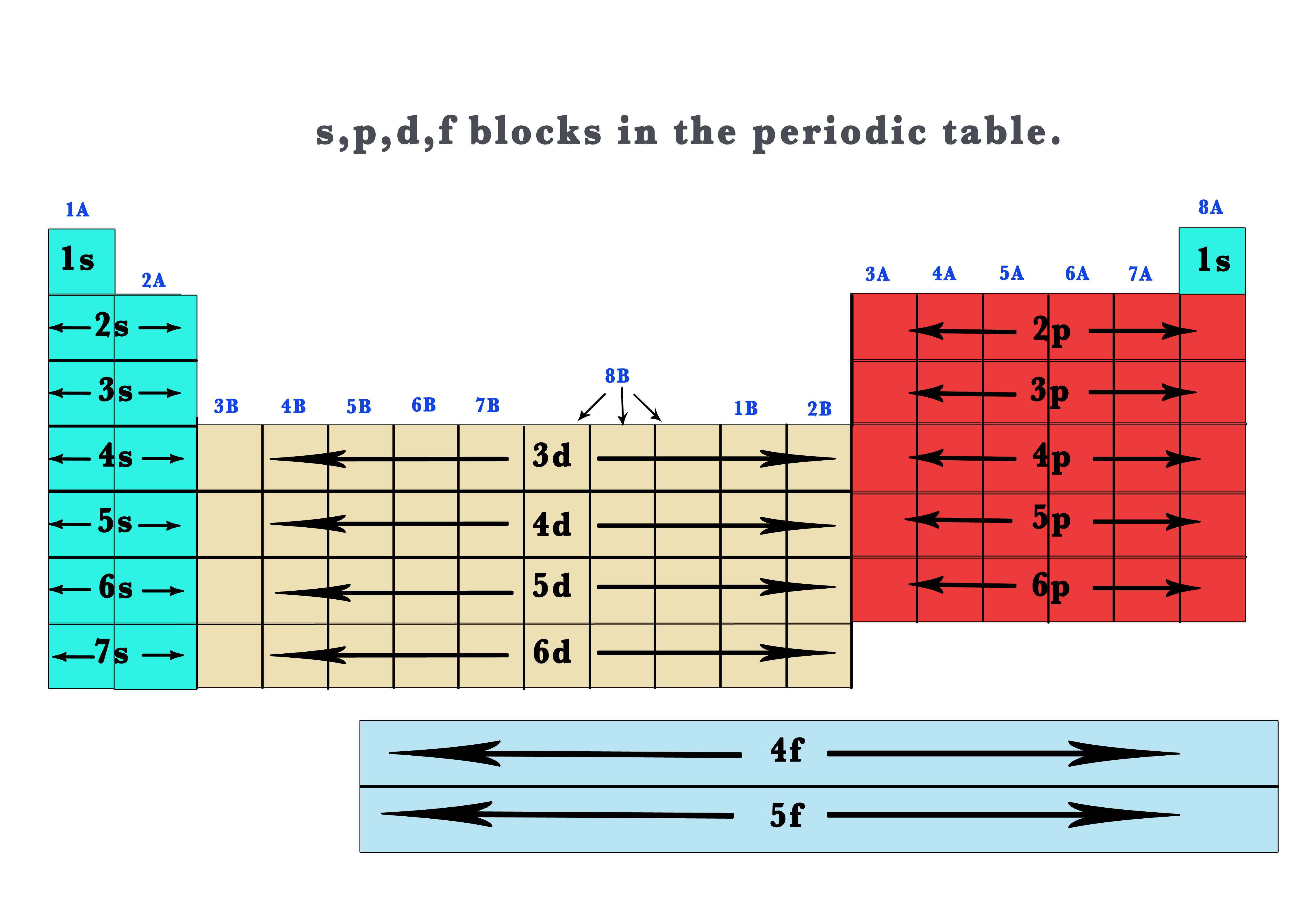


41 The Periodic Table S P D F Blocks Madoverchemistry Com


Parsing The Spdf Electron Orbital Model


Visualizing Electron Orbitals


Electron Configurations How To Write Out The S P D F Electronic Arrangements Of Atoms Ions Periodic Table Oxidation States Using Orbital Notation Gce A Level Revision Notes


Spdf Orbitals Chart Drone Fest



Chm122 2 5 11 Spdf Block Elements Youtube


7 Ways To Color The Periodic Table Students Learn Faster And Retain More



Block Periodic Table Wikipedia


Electron Configuration Chemistry 10
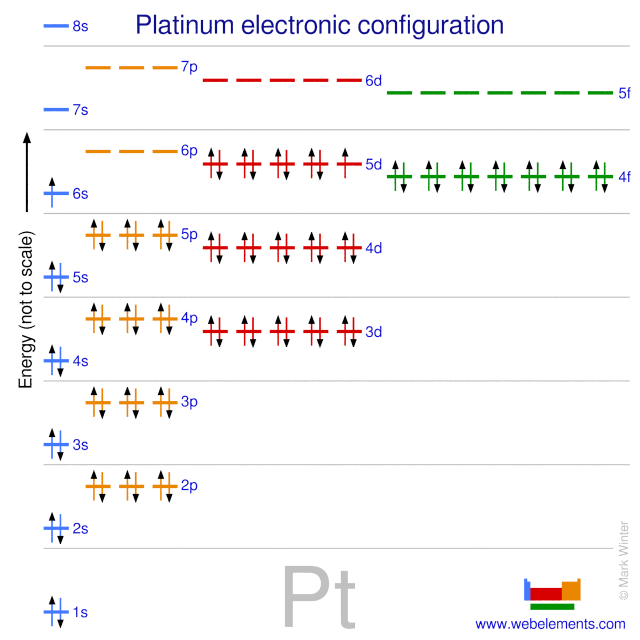


Webelements Periodic Table Platinum Properties Of Free Atoms



What Are Element Blocks On The Periodic Table



Periodic Table Download Periodic Table Pdf Groups Element Names List
/ecblocks-56a129535f9b58b7d0bc9f2e.jpg)


What Are Element Blocks On The Periodic Table



Electron Configuration Boundless Chemistry



Ninth Grade Lesson Introduction To Electron Orbital Levels



A Chart Of The Spdf Electron Orbitals Chemistry Education Chemistry Classroom Teaching Chemistry



Dublin Schools Lesson Electron Configurations Using The Periodic Table


The Order Of Filling 3d And 4s Orbitals Chemistry Libretexts


S P D F Electron Blocks On The Periodic Table Chemistry Periodic Table Electron Configuration Showme


Electron Configurations


コメント
コメントを投稿